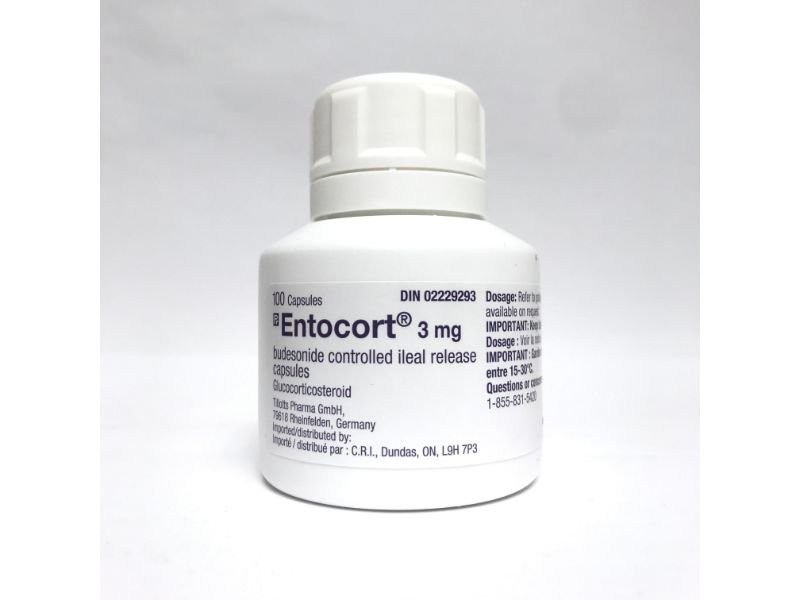
Entocort– Budesonide Oral Therapy for Crohn’s Disease
Entocort delivers targeted relief for patients with mild to moderate Crohn’s disease affecting the ileum and ascending colon. This oral corticosteroid acts locally in the gut, reducing inflammation while minimizing systemic exposure. Patients experience symptom improvement including reduced abdominal pain, decreased frequency of diarrhea, and enhanced overall gastrointestinal function.
Doctors prescribe Entocort for induction and maintenance of remission in Crohn’s disease. Its unique formulation releases budesonide directly at the inflamed site, providing rapid therapeutic action. Patients tolerate the therapy well compared to traditional systemic corticosteroids. Entocort maintains intestinal mucosa integrity, reduces flare-ups, and supports patient quality of life.
Patients take Entocort capsules once daily, with or without food. Capsules release budesonide in a controlled manner, ensuring maximum efficacy in the target region. Regular monitoring ensures optimal results. The therapy supports long-term management plans for Crohn’s disease. Patients benefit from a convenient oral regimen instead of injections or systemic steroids that cause more side effects.
Entocort
Clinical studies show Entocort achieves high rates of remission in mild to moderate Crohn’s disease. Its safety profile remains favorable with minimal systemic corticosteroid effects. Gastroenterologists value its precision-targeted delivery and consistent therapeutic outcomes. Patients report improved energy, reduced gastrointestinal discomfort, and enhanced daily functioning.
Entocort also supports combination therapy strategies. Doctors may integrate it with other maintenance medications to manage disease progression. Patients follow prescribed dosing schedules for maximum benefit. Consistent use ensures inflammation stays under control, preventing complications and hospitalizations.
The formulation consists of enteric-coated capsules containing budesonide. Each capsule ensures uniform drug release along the small intestine. Patients gain predictable dosing and reliable absorption. Entocort’s design emphasizes gut-specific activity, reducing systemic exposure and minimizing typical steroid-related adverse effects.
Product Specifications
| Specification | Details |
|---|---|
| Active Ingredient | Budesonide |
| Formulation | Enteric-coated oral capsule |
| Strength | 3 mg per capsule |
| Indication | Mild to moderate Crohn’s disease (ileum & ascending colon) |
| Administration | Oral, once daily |
| Mechanism of Action | Locally reduces gut inflammation |
| Duration of Therapy | Induction: up to 8 weeks, Maintenance: as prescribed |
| Storage Conditions | Store at 20–25°C, protect from moisture |
| Common Side Effects | Headache, nausea, abdominal discomfort |
| Prescription Requirement | Prescription only |
Entocort empowers patients and doctors to manage Crohn’s disease effectively. Its precision, convenience, and favorable safety profile make it a cornerstone in gastrointestinal therapy.



Reviews
There are no reviews yet.